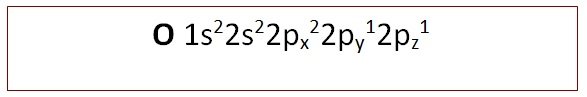 
For instance, most flammable substances are characterised by an autoignition temperature above which they will undergo combustion in air without an external flame or spark. At higher temperatures, or in the presence of suitable catalysts, reactions proceed more readily. Reactions of triplet dioxygen are restricted by conservation of spin state rules and at ambient temperatures this prevents direct reaction with all but the most reactive substrates, e.g. Because of differences in their electron shells, singlet and triplet oxygen differ in their chemical properties. The other singlet state at 157.8 kJmol -1 shown in c) is very short lived and relaxes quickly to b). In isolation, singlet oxygen can persist for over an hour at room temperature. Singlet oxygen is the name commonly used for the electronically excited state shown in b) and it is less stable than the normal triplet state a) by 94.7 kJmol -1. a spin triplet.Ī) e -s parallel (triplet 3Σ g), b) 2 e -s in 1 orbital (singlet 1Δ g), c) e -s opposed (singlet 1Σ g) Note that the spin multiplicity is given by the formula, 2S+1 and so for S=1 from s=½ + ½ then 2S+1 = 3 i.e. With 2 electrons to be placed in 2 degenerate orbitals, a number of variations are possible and the arrangement above where the 2 electrons are parallel is considered to be the most stable. The bond length is 121 pm and the bond energy is 498 kJmol -1.Ī video clip showing liquid dioxygen being poured between the faces of a magnet and attracted into the magnet field has been prepared as a Harvard Natural Sciences Lecture Demonstration. The difference though is that the Lewis structure does not predict the molecule to be paramagnetic. (σ 2s) 2 (σ 2s *) 2 (σ 2p) 2 (π 2p) 4 (π 2p *) 2 and from this the Bond Order is found to be ½(2 -2 + 2 + 4 -2 ) = 2 that is, a double bond as shown in the Lewis structure as well. The electron configuration (ignoring 1s orbitals) is: This is a classic example of where a simple Lewis structure fails to account for the properties and where an MO approach correctly provides the explanation. The electron configuration of O 2 molecules in this form, a diradical, indicates that they should be paramagnetic. 
At STP, dioxygen is a colourless, odourless gas, in which the two oxygen atoms are chemically bonded to each other giving rise to two unpaired electrons occupying two degenerate molecular orbitals. Elemental oxygen is most commonly encountered in this form, as about 21% (by volume) of the Earth's atmosphere is O 2, the remainder largely being dinitrogen, N 2. The common allotrope of elemental oxygen is often just called oxygen, O 2, but to help distinguish it from the element may be called dioxygen or molecular oxygen.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
